- TOP
- Handy Guide of Stainless
- Properties of stainless
Properties of stainless
Stainless steel has been known as less rusty steel as its name shows.
The property of stainless has been due to its oxide film that covers stainless surface.
This oxide film has generally been called as passivate film that is created when chrome, being ingredient of stainless, is exposed and oxidized by the oxygen in the air.
That is why the film can regenerate in the air, even if the film gets any damage by chance.
Since the stainless steel has been covered by this remarkable film, stainless steel retains the superior corrosion resistance, in other word hard to be corroded.
Stainless steel is a general term of the steel of ferrous metal being superior in corrosion resistance that contain about 11% and more chromium and in order to improve its corrosion resistance and mechanical feature, alloy element such as nickel and molybdenum are to be added.
Then highly protective film called passivate film is formed on the surface.
This passivate film is an oxide compound (hydroxide) that contains very thin iron and chromium and the film is extremely thin as thin as being below one hundredth of 1μ.
Therefore, though the film is very vulnerable mechanically and base naked metal will easily be exposed, the film will soon be recovered by the contact with oxygen and vapor and thus regains the corrosion resistance.
- The image of oxide film recovery
![[The image of oxide film recovery]](https://www.abel-s.co.jp/en/wp-content/themes/abel_2020/img/stainless/img1.jpg)
- Composition of
oxide film![[Composition of oxide film]](https://www.abel-s.co.jp/en/wp-content/themes/abel_2020/img/stainless/img2.jpg)
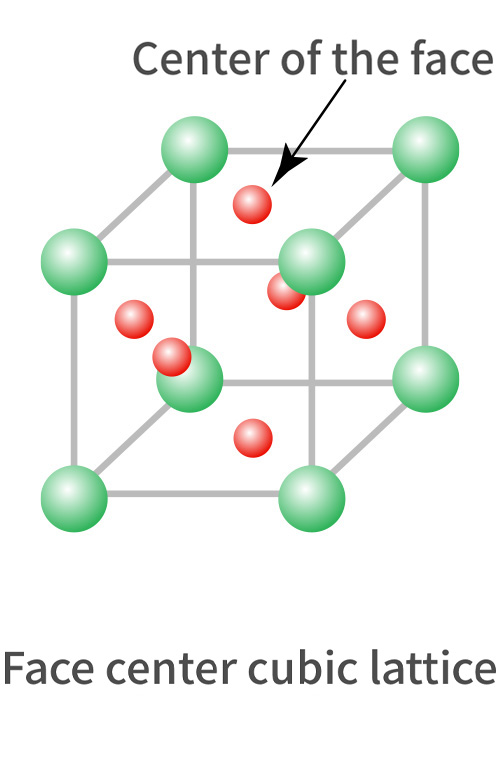
Range of stainless

| Austenite group 18%Cr-8%Ni (SUS304) |
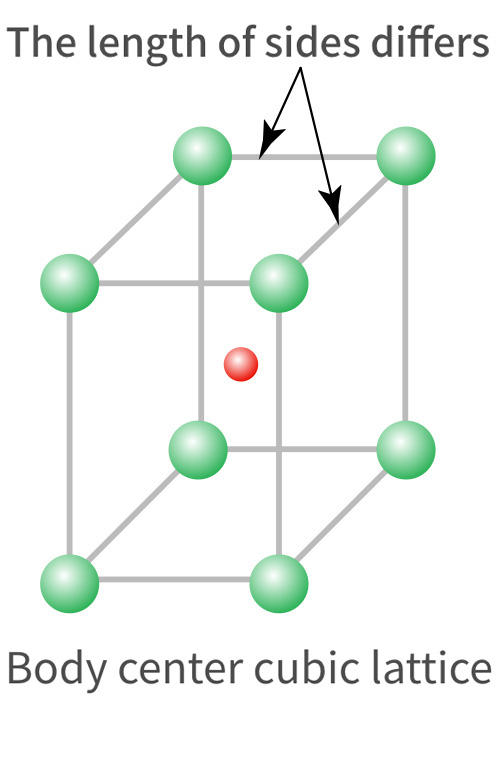
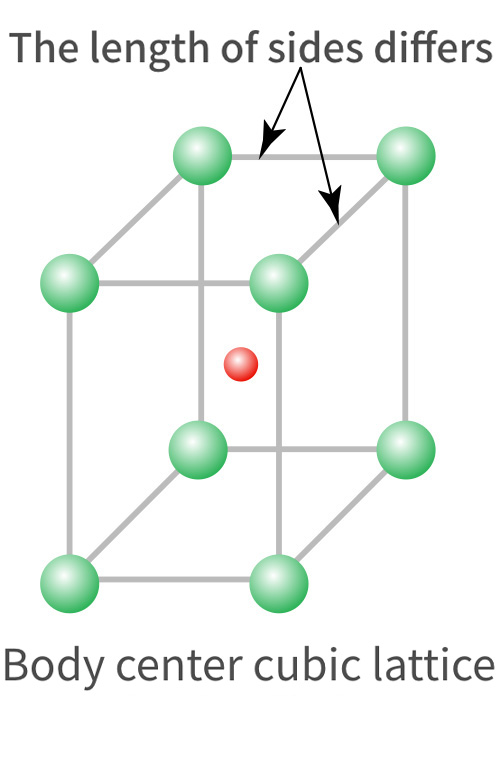
Ferrite group 18%Cr (SUS430) |
Martensite group 12%Cr (SUS410) |
|---|---|---|
 |
 |
 |
| 304 group | 316 group | 430 group |
|---|---|---|
| ・High strength stainless steel
・Ability of press forming ・Grain boundary corrosion resistance
・Stress corrosion breakage resistance
・Oxidation resistance |
・Hole corrosion resistance
・Acidic resistance |
・Heat resistance, oxidation resistance
・Forming, welding
・Corrosion resisting |
Magnetism of stainless
Magnetism ・・・・・there are things that is attracted to a magnet or not.
18-8 (SUS304) is not attracted to a magnet but 18Cr (SUS430) is.
But SUS304 becomes attracted to a magnet if processed.
This is caused by the afore mentioned difference of the crystal structure.
Austenite group are not attracted to a magnet but ferrite group and martensite group are attracted to a magnet.
When SUS304 is given a processing, a part of Austenite construction changes into Martensite system, and result of this it becomes attracted to a magnet.
Stainless has been developed in Germany, England the U.S.A. almost contemporaneously through the year 1912 to 1914, since then the development of various quality of stainless has been progressed.
Various uses of stainless may be developed together with the progress of further technology development in the time to come.



